Crescent Pharma Limited is recalling one batch of Ramipril 5 mg Capsules as a precautionary measure due to a potential error at the manufacturing site.
DMRC Number: DMRC- 38717889
Company name:Crescent Pharma Limited
Product description: Ramipril 5 mg Capsules - PL: 20416/0296, SNOMED code: 38739511000001103, GTIN: 05017123077135
Details of the affected batch:
| Batch No. | Expiry Date | Pack Size | First Distributed |
| GR164099 | 10/2026 | 28 | 06 June 2025 |
Background information:
Crescent Pharma Limited is recalling one batch of Ramipril 5 mg Capsules as a precautionary measure due to a potential error at the manufacturing site. Crescent Pharma Limited has received one complaint to date, where it has been identified that a pack of Ramipril 5 mg Capsules (Batch Number GR164099) contains blister strips of Amlodipine 5 mg Tablets inside the sealed carton. Both products were manufactured at the same manufacturing site and the error appears to have occurred during secondary packaging of the blister strips into the cartons. Please note this is a Class 2 Patient, Pharmacy and Wholesaler level recall.
Advice to Healthcare Professionals:
Stop supplying the impacted batch of Ramipril 5 mg Capsules immediately. Quarantine all remaining stock and return it to your supplier using your approved process.
If batch/product traceability information is available, pharmacy professionals and other healthcare professionals involved in dispensing medicinal products should identify and immediately contact all patients who have been dispensed the impacted product and ask them to confirm if they have remaining stock within their possession for return.
If batch/product traceability information is not available, pharmacists should identify all patients dispensed this product between 06 June 2025 and 06 March 2026. Where appropriate and feasible, contact all patients who have been dispensed the impacted product with priority given to those who have been dispensed the product most recently and within the last 28 days. The majority of this was distributed in 2025 and therefore it is expected that many of these packs will have been dispensed to patients and consumed.
If any patients are identified with this product, pharmacy professionals and other healthcare professionals involved in dispensing should also contact the patients’ GP, or healthcare professional responsible for the care of the patient, to discuss treatment review and if a new prescription is required for ongoing resupply.
Ramipril and amlodipine are both standard anti-hypertensives used in the treatment of hypertension and heart failure. Healthcare professionals should be advised to inform patients who may have taken the incorrect medication that they may experience dizziness symptoms related to blood pressure lowering secondary to taking a calcium channel blocker that they are new to. Additionally, as some patients may require monitoring, other clinicians and healthcare professionals may need to be involved. Healthcare professionals should be aware that only the batch stated in this notification is impacted.
Advice to Patients:
Some cartons of Ramipril 5 mg Capsules, manufactured by Crescent Pharma Limited, may contain blister strips of Amlodipine 5 mg Tablets. All packs of Ramipril 5 mg Capsule, Batch Number GR164099 are being recalled as a precautionary measure.
If you were prescribed Ramipril 5 mg Capsules and have received the impacted product batch (Batch Number GR164099) please check that the carton contains the correct medication. The batch number and expiry date information can be found on outer carton.
- If the carton contains blister strips that are labelled and contain Amlodipine 5 mg tablets, contact your dispensing pharmacy in the first instance. If the carton contains blister strips that are labelled and contain Ramipril 5 mg Capsules, you do not need to take further action.
- If you are unsure or have any questions, please seek advice from your pharmacy or other healthcare professionals responsible for your care.
If you have an impacted pack or previously received this batch, (where known) and you believe you have taken the Amlodipine 5 mg Tablets that were included in error and are currently experiencing any side effects, then please seek immediate medical advice.
Please take the leaflet that came with your medicine and any remaining tablets with you to your pharmacy or GP practice.
Both medications are used to treat high blood pressure, however because your body may not be used to a different type of medicine, your blood pressure may become lower than normal and you may experience dizziness as a result of taking amlodipine.
For reference the description of the products from the Patient Information Leaflets (PILs) are as follows:
- Ramipril 5 mg Capsules: Ramipril 5 mg Capsules are light grey and green gelatine capsules, marked with “R” on the cap and “5” on the body. Click here for PIL
- Amlodipine 5 mg Tablets: Amlodipine 5 mg tablets are white to off white, round, tablets with “5” debossed on one side and plain on the other side – Click here for PIL
See below images showing the different medicines and where to find the batch number of the product and the identification of the incorrect blister strip.
Ramipril 5 mg Capsules – Batch GR164099 Outer carton and BN/Exp Date information
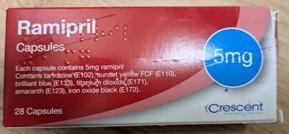

Amlodipine 5mg Tablets – Batch GA094034 Incorrect blister strip

Patients who experience adverse reactions or have any questions about the medication should seek medical attention. Any suspected adverse reactions should also be reported via the MHRA Yellow Card scheme.
Company contacts for further information:
For all medical information enquiries and information, please email medinfo@crescentpharma.com, for reporting of side effects email safety@crescentpharma.com or telephone +44 1217901596 and for stock control enquiries please email complaints@crescentpharma.com
To access the full recall/alert: Class 2 Medicines Recall: Crescent Pharma Limited, Ramipril 5mg capsules, EL(26)A/11